The shift toward gentler isolation
EV labs are moving away from the rotor toward Size Exclusion Chromatography (SEC). The reason: SEC preserves EV biophysical integrity and functionality more effectively than ultracentrifugation (UC), which subjects vesicles to high gravitational forces that can cause aggregation, membrane damage, and reduced biological activity.1-4
In the table below, we compare SEC to Differential Ultracentrifugation (DUC) and Density-Gradient Ultracentrifugation (DGUC).1-6 SEC provides a good balance between yield, purity, and scalability without requiring any expensive instrumentation, making it accessible to most labs. DGUC sets the standard for purity; however, we show that SEC can be improved to address drawbacks of size-only separation methods.
Method comparison
| Parameter | Differential UC (DUC) | Density-Gradient UC (DGUC) | Size-Exclusion Chromatography (SEC) |
| Separation principle | Sedimentation by size/density | Buoyant density in gradient medium | Hydrodynamic radius (size) |
| EV yield | Low–Moderate | Low | High |
| Protein removal (purity) | Low–Moderate | High | Moderate–High |
| Lipoprotein depletion | Poor | High separation from low-density lipoproteins | Poor for low-density lipoproteins (SEC); High (multi-mode SEC) |
| EV integrity / functionality | Reduced (aggregation risk) | Moderate (long processing) | High |
| Processing time per run | 4–18 hours | 16–72 hours | ~30 minutes |
| Scalability / throughput | Low | Low | High (parallelizable, automatable) |
| Equipment | Ultracentrifuge | Ultracentrifuge | SEC column, automation instrument optional |
SEC with Apex columns
The Apex SEC columns are optimized to deliver a reproducible EV elution profile by validating each lot for consistent drip speed (Read more) and verifying elution by Atlas EV ELISA. Apex 6B columns are packed with Sepharose CL-6B and are optimized for the highest EV recovery. Apex 6B is perfect for separating EVs from soluble proteins in less complex samples (e.g. cell culture, CSF and urine). If you’re working with demanding samples like plasma and serum, Apex 4B columns are packed with Sepharose CL-4B and optimized for the highest purity of secreted proteins.
The low-density lipoprotein challenge & Apex MM solution
MISEV2023 highlights that no single separation method fully resolves EVs from co-isolating non-vesicular extracellular particles (NVEPs) and recommends that researchers select and combine methods based on the specific requirements of their downstream application.5 The separation of EVs from NVEPs is most challenging when isolating EVs from serum or plasma since lipoproteins are several orders of magnitude more abundant than EVs. These difficulties are present in both UC and SEC, except when using laborious Density-Gradient UC.
Apex MM columns address this challenge by combining SEC resin with a small pore size and a multi-mode (MM) resin (Figure 1). This resin utilizes hydrophobic interactions and ion-exchange to selectively bind and remove lipoproteins and other soluble contaminants. Apex MM columns are particularly advantageous for unbiased downstream analyses, such as proteomics, where purity is paramount.
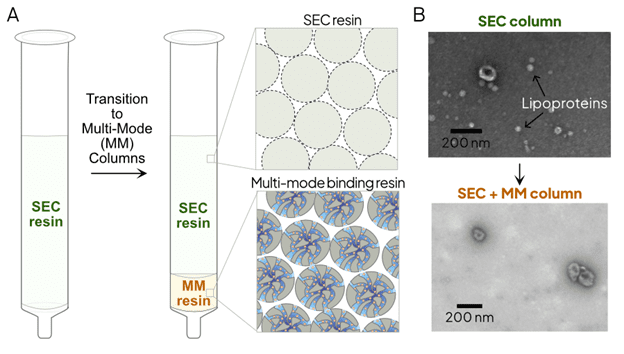
Figure 1. Apex MM column architecture.
A. Schematic representation of the transition from standard SEC to multi-mode chromatography. The integrated MM resin layer provides a secondary purification step by binding lipoproteins and soluble proteins through hydrophobic and ionic interactions.
B. TEM images of EVs isolated using SEC column (top) and Apex MM column (bottom).
Apex MM improves SEC to provide reproducible and pure EV preparations for the most demanding samples, providing users with a reproducible and automatable workflow versus DG UC.
SEC automation for scalable processing
The Ascent instrument enables semi-automated SEC-based EV isolation for up to 8 samples simultaneously, while the Summit instrument scales this fully automated EV isolation from 1 up to 48 samples at a time. By eliminating operator-dependent variability, these instruments support the throughput required for biomarker discovery and clinical research, effectively addressing the key historical limitation of manual SEC.
Solving the EV concentration challenge through automated ultrafiltration
For certain applications, SEC dilution of EVs can limit downstream analyses requiring low input volumes. To solve this, SEC fractionation can be coupled with ultrafiltration (UF) to deliver concentrated EV fractions. This workflow is easily implemented on the Ascent instrument by collecting EVs directly into centrifugal concentrators (Read more). For larger cohorts, the Summit instrument provides a fully automated, plate-based format that integrates both SEC isolation and UF concentration for up to 48 samples in parallel—offering a high-throughput, hands-free solution for large-scale discovery studies (Explore the Summit Instrument).
Summary
The transition from ultracentrifugation (UC) to Size-Exclusion Chromatography (SEC) represents a fundamental shift toward biological integrity and laboratory scalability. The Apex SEC columns are optimized for consistent and reproducible EV isolation. While traditional SEC once struggled with lipoprotein co-isolation and sample dilution, the advent of Apex MM columns and automated concentration workflows has provided a robust solution. These advancements maintain the inherent gentleness of SEC while achieving the high purity required for advanced proteomics.6 When paired with the Ascent and Summit platforms, these workflows move EV isolation out of the “expert-only” realm of manual ultracentrifugation and into a standardized, high-throughput future.
References:
- Mol EA, Goumans MJ, Doevendans PA, Sluijter JPG, Vader P. Higher functionality of extracellular vesicles isolated using size-exclusion chromatography compared to ultracentrifugation. Nanomedicine, 2017
- Linares R, Tan S, Gounou C, Arraud N, Brisson AR. High-speed centrifugation induces aggregation of extracellular vesicles. J Extracell Vesicles, 2015
- Nordin JZ, Lee Y, Vader P, Mäger I, Johansson HJ, Heusermann W, Wiklander OP, Hällbrink M, Seow Y, Bultema JJ, Gilthorpe J, Davies T, Fairchild PJ, Gabrielsson S, Meisner-Kober NC, Lehtiö J, Smith CI, Wood MJ, El Andaloussi S. Ultrafiltration with size-exclusion liquid chromatography for high yield isolation of extracellular vesicles preserving intact biophysical and functional properties. Nanomedicine, 2015.
- Cvjetkovic A, Lötvall J, Lässer C. The influence of rotor type and centrifugation time on the yield and purity of extracellular vesicles. J Extracell Vesicles, 2014
- Welsh JA, Goberdhan DCI, O’Driscoll L, Buzas EI, Blenkiron C, Bussolati B, Cai H, Di Vizio D, Driedonks TAP, Erdbrügger U, Falcon-Perez JM, Fu QL, Hill AF, Lenassi M, Lim SK, Mahoney MG, Mohanty S, Möller A, Nieuwland R, Ochiya T, Sahoo S, Torrecilhas AC, Zheng L, Zijlstra A, Abuelreich S, Bagabas R, Bergese P, Bridges EM, Brucale M, Burger D, Carney RP, Cocucci E, Crescitelli R, Hanser E, Harris AL, Haughey NJ, Hendrix A, Ivanov AR, Jovanovic-Talisman T, Kruh-Garcia NA, Ku’ulei-Lyn Faustino V, Kyburz D, Lässer C, Lennon KM, Lötvall J, Maddox AL, Martens-Uzunova ES, Mizenko RR, Newman LA, Ridolfi A, Rohde E, Rojalin T, Rowland A, Saftics A, Sandau US, Saugstad JA, Shekari F, Swift S, Ter-Ovanesyan D, Tosar JP, Useckaite Z, Valle F, Varga Z, van der Pol E, van Herwijnen MJC, Wauben MHM, Wehman AM, Williams S, Zendrini A, Zimmerman AJ; MISEV Consortium; Théry C, Witwer KW. Minimal information for studies of extracellular vesicles (MISEV2023): From basic to advanced approaches. J Extracell Vesicles. 2024
- Guan, X., Liu, J., et al. (2022). Small extracellular vesicles isolation and separation: Current techniques, pending questions and clinical applications. Theranostics, 12(15), 6548-6574.
- Ter-Ovanesyan D, Gilboa T, Budnik B, Nikitina A, Whiteman S, Lazarovits R, Trieu W, Kalish D, Church GM, Walt DR. Improved isolation of extracellular vesicles by removal of both free proteins and lipoproteins. Elife. 2023